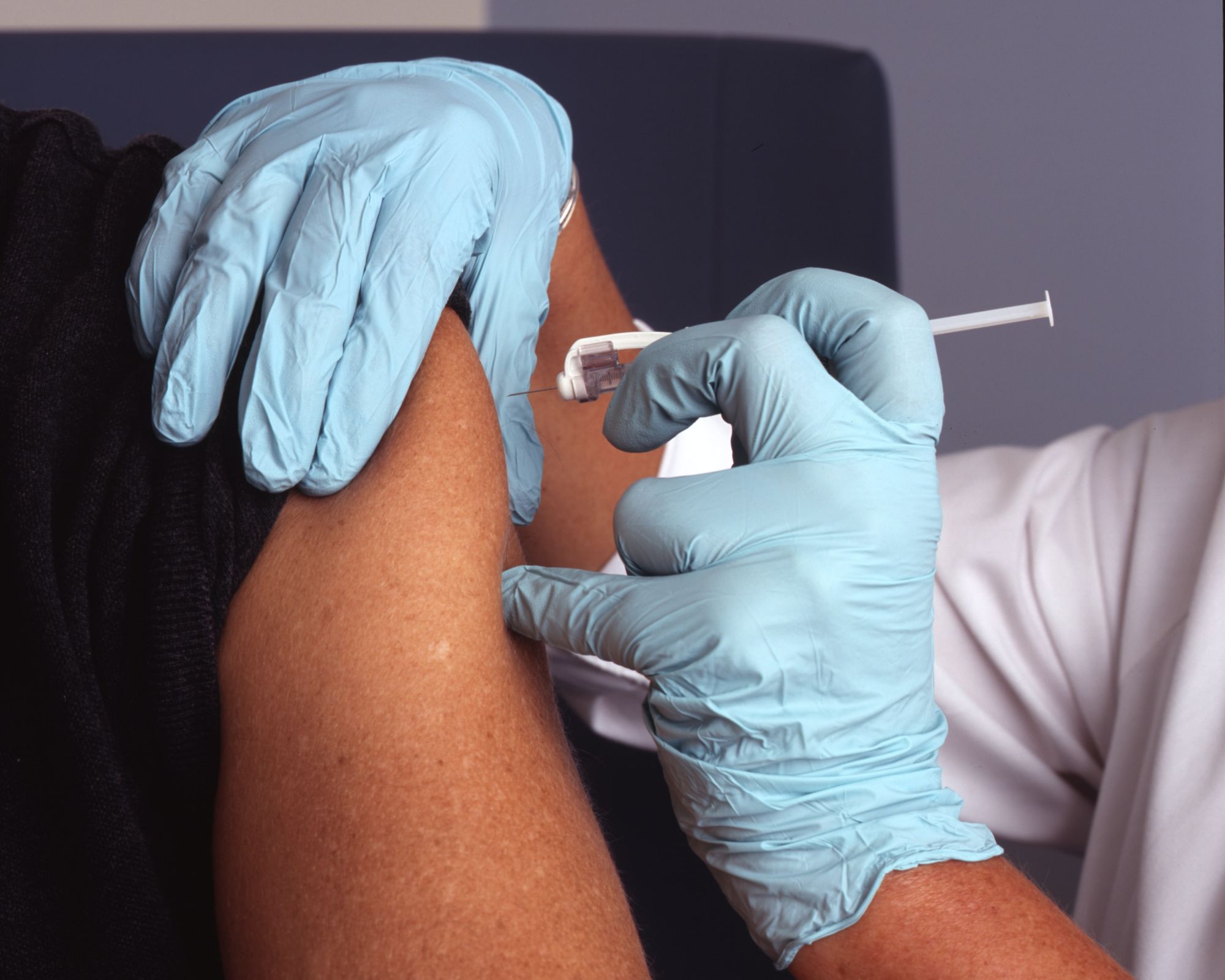
The first Canadian clinical trials for a potential COVID-19 vaccine are set to begin.
The trials will be conducted by the Halifax research team that was involved in the eventual Ebola virus vaccine in 2014. They will take place at the Canadian Centre for Vaccinology at Dalhousie University.
While releasing a vaccine usually takes anywhere from five to seven years, there is a strong chance the process will be expedited given the circumstances. Six years ago, a vaccine was tested on Ebola patients in West Africa after around half a year of trials. It wasn’t until last year that the Ebola vaccine was licensed as a regular marketed vaccine.
The speed at which a possible COVID-19 vaccine will be made available to the public depends on Health Canada and the Canadian government.
And, of course, its efficacy.
A Chinese manufacturer for the vaccine is already conducting human clinical trials for the vaccine. The first trial in Canada will take place in the next three weeks. Fewer than 100 healthy volunteers between the ages of 18 and 55 will participate in the first phase of the trial. They will then be evaluated over the course of six months.
If participants in the first phase show signs of immunity, the second phase will begin even before the first is over. The second phase will involve hundreds of people of all ages, including high-risk groups.
If the vaccine shows promise in the second phase, the Halifax lab will begin working with Canadian health officials to expedite the process of making it available to the public.